STATRIX – Hemostatic Gel
₹980.00
Fast-acting hemostatic gel for effective bleeding control during restorative and surgical procedures. STATRiX offers high viscosity for precise placement, rapid hemostasis, and gentle tissue response.
STATRiX Hemostatic Gel is a fast-acting, high-viscosity gel formulated for rapid bleeding control and effective soft-tissue management during restorative, periodontal, and minor surgical procedures. Designed to stay precisely where applied, it provides reliable hemostasis while remaining gentle on gingival tissues.
Its syringe-based delivery system allows controlled placement, making it especially effective in difficult-to-access areas. STATRiX Gel helps create a clean, dry working field, improving visibility, procedural accuracy, and overall clinical efficiency.
Easy to rinse and well tolerated by tissues, STATRiX supports optimal post-operative tissue health while reducing chairside time—making it an essential consumable for everyday dental practice.
Indications
-
Control of minor gingival bleeding during restorative procedures
-
Use before impression taking or bonding procedures
-
Hemostasis during crown preparation or matrix band placement
-
Soft tissue management in surgical and scaling procedures
Key Advantages
-
Rapid hemostasis
-
High-viscosity gel for controlled placement
-
Easy to rinse off
-
Gentle on soft tissues
-
Syringe delivery for precision
-
Enhances overall procedure efficiency
Why Choose STATRiX Gel?
-
Faster bleeding control compared to conventional hemostatic gels
-
Minimal tissue irritation and improved patient comfort
-
Effective even in difficult clinical areas
-
Supports excellent post-operative tissue health
STATRiX delivers dependable bleeding control—control at your fingertips.
Packaging:
3 g × 1 syringe
Related products
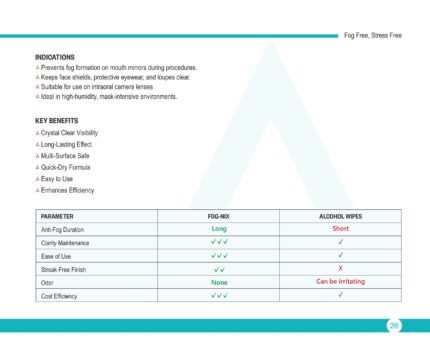
FOG-NIX – Anti-Fog Spray
GLANZAA – Diamond Polishing Paste
LINICAL – Light Cure Calcium Hydroxide Liner
MARK X – Caries Indicator Dye
MG3 Blue
MG3 Gold is a premium gold heat-treated rotary endodontic file system designed for safe, efficient, and predictable root canal shaping. Engineered for enhanced flexibility and high resistance to cyclic fatigue, it allows smooth canal preparation while maintaining original canal anatomy. Ideal for curved and complex canals, MG3 Gold delivers controlled cutting performance with reduced risk of file separation, making it a reliable choice for modern endodontic practice.
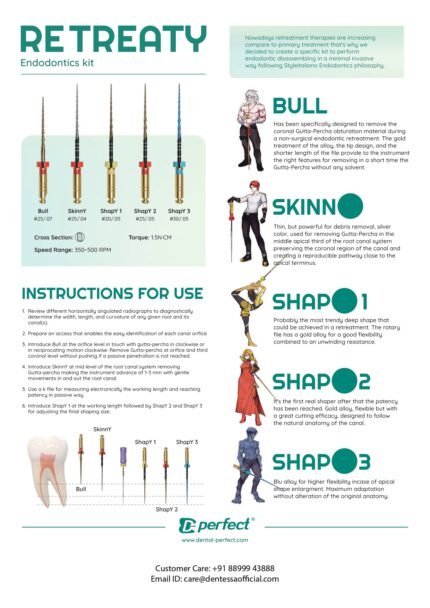
RETREATY – Endodontics Kit
RETREATY is a purpose-built endodontic retreatment kit designed for efficient, non-surgical removal of gutta-percha without solvents. Featuring a sequential system of rotary files—BULL, SKINNY, and SHAPY 1–3—it enables controlled coronal, middle, and apical retreatment while preserving the natural canal anatomy. Operating at 350–500 RPM with 1.5 N·cm torque, RETREATY delivers safe, minimally invasive retreatment with predictable clinical results.
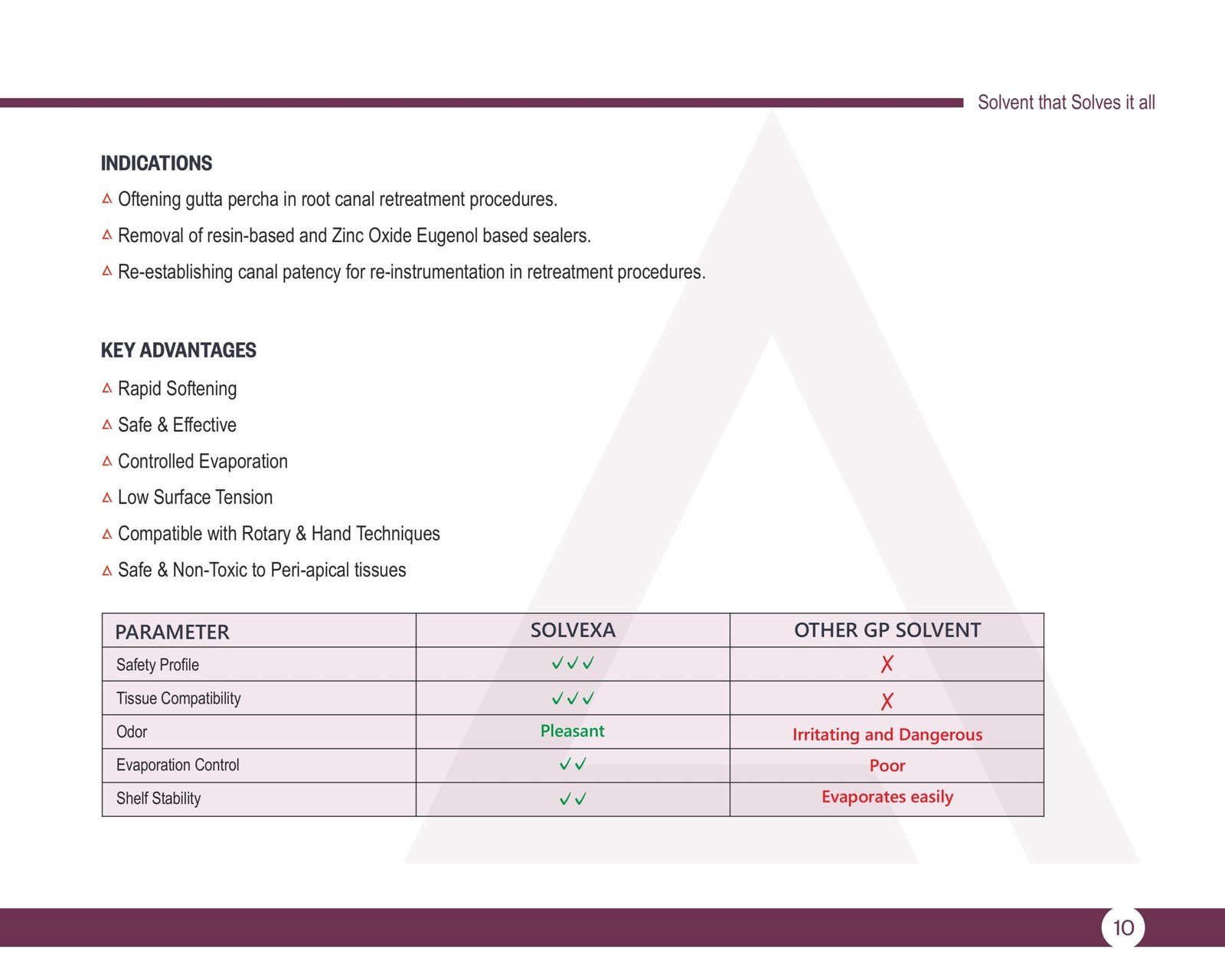
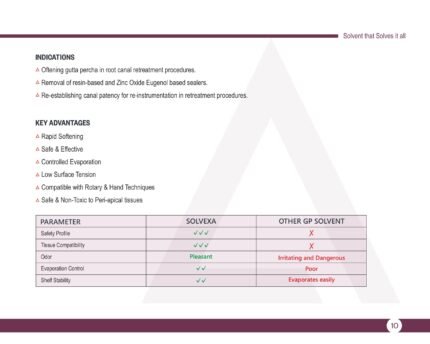
SOLVEXA – Gutta Percha Solvent
TS4
TS4 is a heat-treated NiTi rotary endodontic file system engineered for safe, flexible, and efficient root canal preparation. Featuring a unique W-shaped cross section, it minimizes dentinal stress and reduces the risk of micro-crack formation while delivering excellent cutting efficiency and resistance to cyclic fatigue. With multiple size options, controlled torque requirements, and smooth performance at both low and high rotational speeds, TS4 allows precise canal shaping while preserving the natural anatomy of the root canal system.





















Reviews
There are no reviews yet.